

2020-05-27
PREFACE: Single-base gene editing technology, which has attracted much attention since it was reported in April 2016 for not introducing DNA double-strand breaks and for more efficient and safe editing, has gradually become one of the popular tools for gene therapy of genetic diseases such as thalassemia and hemophilia in recent years. However, existing base editors can only catalyze a single type of base C or A, which limits their wide application for a longer period of time and to a large extent. Therefore, developing a simple and efficient base editor to realize two different types of base conversions, i.e., C toT and A toG simultaneously, has been the focus and difficulty of base editor optimization exploration.
BRL Medicine Inc., in collaboration with Prof. Liu Mingyao and Prof. Li Dali of East China Normal University, published a research paper entitled "Dual base editor catalyzes both cytosine and adenine base conversions in human cells" in Nature Biotechnology on June 1, 2020. The research paper entitled "Dual base editor catalyzes both cytosine and adenine base conversions in human cells" was published in Nature Biotechnology. This is another major breakthrough in the development of single-base editing tools byBioraylab in 2020, following the gene therapy results published in Nature Medicine, Nature Cell Biology, Cell Research and other journals, providing new directions and tools for basic research and treatment of genetic diseases such as β-thalassemia. Xiaohui Zhang, Biyun Zhu and Liang Chen of East China Normal University are co-first authors of this paper, and Professor Dali Li of College of Life Sciences, East China Normal University is the corresponding author.

In this paper, the researchers fused cytidine (hAID) and adenosine deaminases to Cas9n (SpCas9 D10A mutant) and achieved simultaneous C·G and A·T base editing on the same allele as well as C·G or A·T base editing, creating a new base editor, A&C-BEmax. Compared with ABEmax and AID-BE4max (with optimized codon, NLS and uracil DNA glycosylase inhibitor (UGI) copies), A&C-BEmaxwas able to expand the C-to-T editing window and enhance the C-to-T editing efficiency. The A-to-G editing window of A&C-BEmax was consistent, and the A-to-G editing efficiency was comparable or slightly decreased, and the RNA off-target was greatly reduced.
Unique innovation: realizing C·G and A·T base editing simultaneously
Owing to the fact that CBEs or ABEs only catalyze the conversion of a single type of nucleotide, either C·G-T·A or A·T-G·C, the groups hypothesized that by recombining the tools of CBEs or ABEs, they could accomplish a feasible and efficient conversion of the two types of nucleotides for one time. Five constructs were generated with combinations of the two deaminases and Cas9n and tested in the mammal cells to convince this. Finally, they’ve found cytidine deaminase fused to the N terminus of ABE7.10 induced significant levels of simultaneous A/C mutation in the same allele, but the editing efficiency was relatively low.
Higher editing efficiency by adopting optimized schemes
To perfect thedeficiency of relatively low efficiency, the researchers continuously tried out different experimental designs and adjusted the technical methods, and eventually found that by optimizing the Cas9n codon and the Linker sequence in ABE7.10-N-AID, adding a bipartite nuclear localization signal (bpNLS) and fusing two copies of uracil DNA glycosylase inhibitor (UGI), an efficient A/C simultaneous base editor, named A&C-BEmax (Fig.1), was possessed.
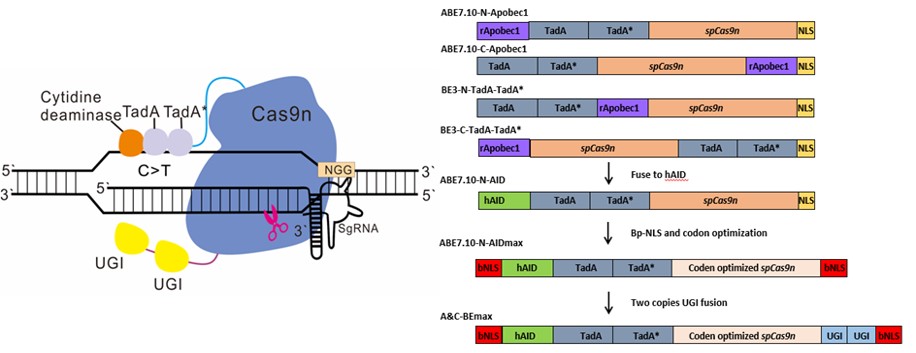
Fig.1 Schematic diagram of A&C-BEmax and its diverse constructions.
A&C-BEmax: A specific dual-function base editor
Compared with the original construction, A&C-BEmax significantly improved the base conversion efficiency, product purity, and A/C simultaneous conversion activity. It greatly increased the editing window of C toT, and was safer for the less RNA off-target. To evaluate the performance of A&C-BEmax more extensively, the researchers tested 28 endogenous targets (four of which contained only Cs or As) in HEK293T cells and compared them with both ABEmax and AID-BE4max.
The results showed that at the same endogenous target, the C>T editing window of A&C-BEmax extended from C3-C10 to C2-C17, and the editing efficiency of C7-C17 was 1.9-14 times higher than that of AID-BE4max (Fig.2). Whereas the editing efficiency of A toG was similar to or slightly lower than that of ABEmax, but the RNA off-target was less than ABEmax, and the simultaneous mutation editing efficiency of A and C of the same allele reached up to 30%. Meanwhile, the data showed that compared with CBE and ABE, A&C-BEmax notably improved editing efficiency, product purity, base mutation saturation and mutational types, and A/C simultaneous conversion activity. The DNA off-target was nearlyundetectable and RNA off-target was also largely reduced. Hence, A&C-BEmax served as a dual-function base editor with high specificity and safety.
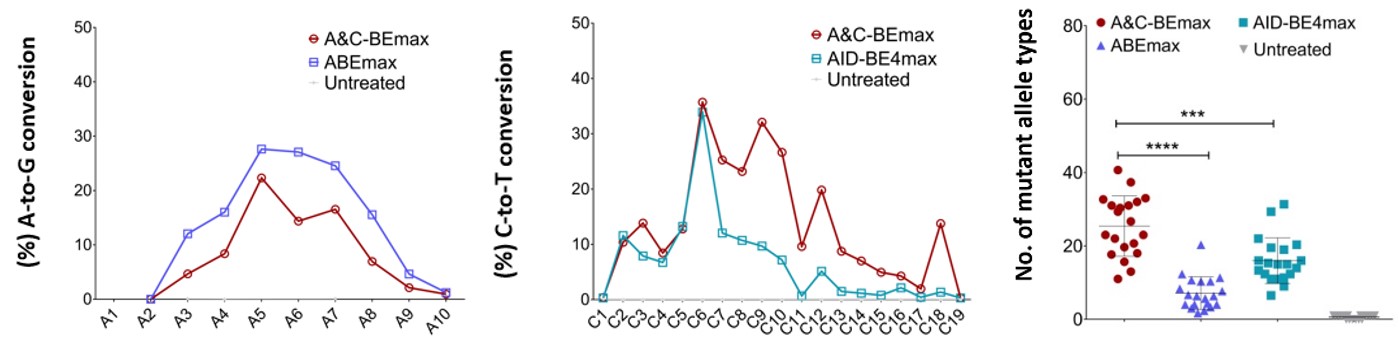
Fig.2 The characteristics of A&C-BEmax
New base editor in thalassemia treatment, shows great potential
The newly developed base editor displays great potential in treating thalassemia
To further verify the application of A&C-BEmax in gene therapy, A&C-BEmax was delivered intoerythroid precursor cell lines(HUDEP-2). The results showed that A&C-BEmax could effectively destroy the binding site of BCL11A transcription suppressor (-114C>T or -115C>T). As a combined effect, the binding site of transcriptional activator GATA1 (-113A>G) was also created, which could not be accessible by existing ABEs or CBEs. Compared with cells solely producingC editing, the differentiated HUDEP-2 cells with simultaneous−113A/−114C conversion induced the highest HBG expression (Fig.3). This experiment showed the huge potential of A&C-BEmax for the effective treatment ofβ-hemoglobindisease, and provides another crucial approach for curing the severeβ0/β0 -thalassemia.
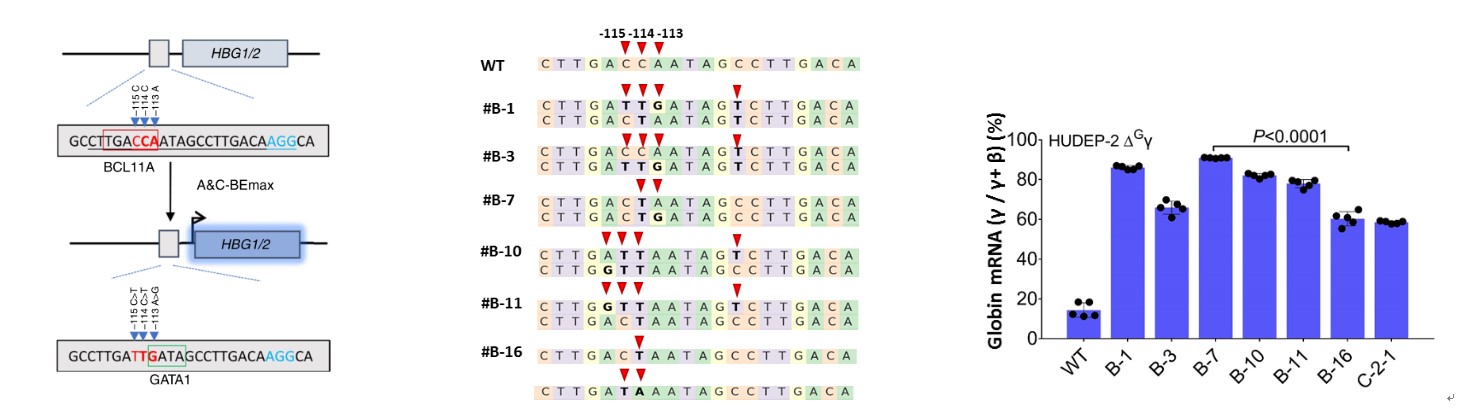
Fig.3 The high activation of HBG expression by A&C-BEmax in HUDEP-2 cells
Finally, the team analyzed all A&C-BEmax targetable by computer in the Clin Var and dbSNP (SNPs) clinical databases to correct 203 clinically reported pathogenic mutations by combining C toT and A toG targeting; the targeting scopeincreased by 2.8-fold (~573) when further expanding PAM to NG. This suggests that A&C-BEmax has great potential for clinical application in the correction of human pathogenic mutations caused by combining C toT and A toG.
BRL Medicine makes breakthroughs in the field of gene therapy
Since its establishment, BRL Medicine has been focusing on gene editing and gene therapy, independently developed and built a gene therapy technologyconversionplatform, and has filed more than 30 global and Chinese patent applications in the field of gene editing and gene therapy. Looking back on the pioneering path of BRL Medicine, it is a process of continuous breakthroughs and innovations, especially since last year, BRL Medicine's team of scientists has continued to make many breakthroughs in the field of gene editing tools development and gene therapy for thalassemia.
In March 2019, BRL Medicine Biosciences scientist Dr. Yuxuan Wu, published an article in Nature Medicine, discovering that targeted editing of the BCL11A red-lineage enhancer using gene editing technology would re-enable γ-bead protein expression in place of the defective β-bead protein, promising a radical cure for such diseases.
In January 2020, the team of Professors Mingyao Liu and Dali Litake the lead in proving the feasibility and effectiveness of single-base editor editing technology targeting the HBG promoter to activate hemoglobin expression in fetal life for the treatment of β-thalassemia, and the related research was published in the internationally renowned academic journal Cell Research.
In March 2020, Dr. Yuxuan Wu, a scientist at BRL Medicine, published an article in the prestigious international academic journal Nature Medicine, demonstrating that single-base editing technology can target the red lineage enhancer progenitor of BCL11A to activate fetal hemoglobin or edit specific mutations in the β-hemoglobin gene, promising to treat β-hemoglobin disorders including β-thalassemia by editing autologous hematopoietic stem cells.
In May 2020, the latest article published in Nature Cell Biology also reported that the latest series of hyCBEs tools developed by Prof. Li Dali's team possess higher editing activity and wider editing window, which have greater advantages for the treatment of β-hemoglobinopathies.
This breakthrough research, moreover, is a major research result to break the limitation of existing base editors, develop a new bifunctional base editor with source innovation, and create a domestic independent intellectual property rights gene editing tool, which provides a new platform for basic research and treatment of genetic diseases such as β-thalassemia.
Mr. Zaixi XI, co-founder and president of BRL Medicine, revealed that Bioraylab has built an innovative and efficient gene editing platform, and that Bioraylab's single base editor and its series of breakthroughs in gene therapy for genetic diseases since 2020 will add another tool to gene therapy, and BRL Medicine is focusing on clinical translation around these independent intellectual property technologies. Since its inception, BMI has been focusing on the development of gene and cellular drugs, and has already collaborated with several medical institutions in China to achieve breakthroughs in the research of genetic diseases and malignant tumors. We look forward to bringing benefits to more patients with cancer and genetic diseases in the future.
Zhang, X., Zhu, B., Chen, L. et al. Dual base editor catalyzes both cytosine and adenine base conversions in human cells. Nat Biotechnol (2020).
https://doi.org/10.1038/s41587-020-0527-y